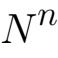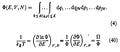Introduction
Our previous post showed that the equilibrium molecular-kinetic-energy gradient a gravitational field imposes on an isolated ideal gas could not in theory be exactly zero even though Fourier’s Law may seem to require otherwise. This post will show that thermodynamics defines that problem away by rejecting the Ideal-Gas Law definition of temperature. It will also give an example of how this definition difference can cause confusion.
Kinetic Energy and Temperature
One form of the Ideal-Gas Law is P = nkT, where P is pressure, n is the number of molecules per unit volume, k is Boltzmann’s constant, and T is absolute temperature. That a (monatomic) ideal gas’s pressure P equals two-thirds of the product of the gas’s molecular density n and mean molecular kinetic energy can readily be derived from first principles. So for an ideal gas the Ideal-Gas Law defines temperature implicitly; temperature has to be a quantity proportional to that kinetic-energy value.
Specifically, the temperature of an ideal monatomic gas is two-thirds of its mean molecular kinetic energy divided by Boltzmann’s constant. This means that in accordance with the Ideal-Gas Law definition the previous post’s demonstration of mean molecular kinetic energy’s decrease with altitude proves that the lapse rate—i.e., the altitude rate of temperature change—would have to be non-zero in a finite gas.
But temperature isn’t a property of ideal gases only, and in the lapse-rate context temperature definitions of wider applicability can yield different results in theory (although in practice the differences would be too small to measure). Specifically, the Zeroth Law of Thermodynamics states that between bodies in thermal communication with one another heat must flow from the hotter to the cooler until their temperatures are equal, and some physicists view that law as an implicit definition of temperature: the temperatures of thermally coupled bodies are equal by definition if heat doesn’t flow between them.
According to this view, temperatures at different altitudes in a gas at equilibrium—i.e., in a gas in which heat flow has died out—are equal by definition even if the mean molecular kinetic energies at those altitudes differ. And the previous post demonstrated that at equilibrium different altitudes’ mean molecular kinetic energies can’t be exactly equal. So the Ideal-Gas Law definition can yield theoretical results that differ, if ever so slightly, from those implied by the Zeroth Law.
Thermodynamics as an Approximation
One way to view the difference is to think of thermodynamics as an approximation, albeit an exquisitely good one. Thermodynamics developed somewhat empirically back during the development of steam engines. The well-known thermodynamics relationship dS = dQ/T among a body’s entropy S, the heat Q that flows into it, and its temperature T was inferred without much reference to the molecules of which the body consists. Moreover, its experimental verification has necessarily been limited by measurement accuracies. And even much of the later-added theoretical support was based on approximation.
Consider, for example, the Maxwell-Boltzmann distribution, on which Coombes & Laue’s zero-lapse-rate demonstration mentioned in the previous post was based. That distribution was in turn based on the Boltzmann distribution, whose derivation often begins by considering a system (not necessarily a gas column) that’s in thermal equilibrium with a high-heat-capacity environment (a “heat bath”).
A good example of such a derivation starts at time stamp 28:27 in a Leonard Susskind statistical-mechanics video. Because of the continual energy exchange between the system and its heat bath the states assumed by the system can have different energy values, and the derivation in the Susskind lecture concerned the probabilities of the system’s assuming respective states.
The fact that for a given heat bath those total energies will have some average over time serves as a constraint on the probabilities’ distribution. And, although there are many probability distributions that could meet that constraint, the one in which the most combinations result is found by maximizing entropy S:
where the i’s label the different states a system can assume and the P’s are those states’ probabilities. Although that equation isn’t intuitively appealing, it may help to note that when all states are equally probable it boils down to a statement that entropy is simply proportional to the number of states.
It can be shown (as Dr. Susskind did) that in the limit as the environment’s heat capacity approaches infinity the system’s entropy is maximized when the states’ probabilities decay exponentially as energy increases:
where the subscripted E’s are the individual states’ energies and β is a coefficient that depends on the assumed average energy (non-subscripted) E.
That distribution results in dS/dE = β. Replacing infinitesimal heat flow dQ in the entropy equation dS = dQ/T with the infinitesimal average-energy change dE in dS/dE = β defines temperature as the reciprocal of kβ, thereby making the above-defined most-likely distribution the Boltzmann distribution.
The point to note about that derivation is that the Boltzmann distribution resulted from taking the limit as the environment’s heat capacity approaches infinity. For a finite system the Boltzmann distribution is merely an approximation. Avogadro’s number being what it is, of course, it’s an exceedingly good approximation. As the previous post explained, however, the thereby-implied vertical-component distribution can’t be exactly exponential if the number of molecules isn’t infinite, so under the Ideal-Gas Law definition the equilibrium lapse rate can’t be exactly zero.
Velasco et Al.’s Lapse-Rate Value
What is the equilibrium lapse rate if it’s not zero? The answer was given in a Velasco et al. letter written in response to Coombes & Laue’s paper. Velasco et al. confirmed that Coombes & Laue’s conclusion—i.e., that the molecules’ mean molecular kinetic energy is independent of altitude—is valid in the infinite limit. But “for a finite adiabatically enclosed ideal gas in a gravitational field,” they wrote, “the average molecular kinetic energy decreases with height.”
Specifically, their value for the mean value <K> of molecular kinetic energy as a function of altitude z is given by:
where f = 3 for a monatomic gas, E is the gas column’s total (potential + kinetic) energy, and N is the number of molecules.
Now, that value underscores how academic the distinction is between zero and nonzero lapse-rate values; for a mole of gas whose atomic weight is the air-average 29 the lapse rate given by that equation would be less than a trillionth of a trillionth of a kelvin per meter: much too small to measure. Even if individual measurements were perfectly accurate, in fact, the rate given by that equation is probably so dwarfed by the lapse rate’s random fluctuations that averaging those fluctuations out enough to detect the underlying rate would require too many measurements to be practical.
A Matter of Definition
Still, the gradient Velasco et al. found is not exactly zero, so in accordance with the Ideal-Gas Law definition the lapse rate isn’t either. But some scientists disagree with that reasoning—because they reject the Ideal-Gas Law definition of temperature. In fact, Velasco et al. themselves seemed to reject the Ideal-Gas Law definition.
Recall in this connection that Coombes & Laue’s paper was an answer to the “paradox” presented by a thermally isolated ideal-gas column at equilibrium in a gravitational field. The paradox was that physics seemed to support two contradictory statements:
(1) There is no temperature gradient because a system which is in equilibrium has the same temperature throughout.
(2) The temperature decreases as one goes up the column for the following reasons. (a) Since . . . every molecule loses kinetic energy as it moves upward, the average kinetic energy of all molecules decreases with height. (b) Temperature is proportional to the average kinetic energy.
As we saw in the previous post, Coombes & Laue argued that reason (2a)—and therefore statement (2)—are wrong by demonstrating that gravitational culling can keep the average kinetic energy independent of height even though every molecule loses kinetic energy as it travels upward.
Velasco et al.’s penultimate paragraph stated as our previous post did that Coombes & Laue’s analysis—i.e., their demonstration that reason (2a) is wrong—is valid only in the limit as the number of molecules approaches infinity. Additionally, however, that paragraph implicitly rejected the Ideal-Gas Law’s definition of temperature: it rejected the reason (2b) proposition that temperature is proportional to average kinetic energy:
In conclusion, in our opinion a full explanation about why answer (2) to the paradox formulated by Coombes and Laue is wrong must discern between the cases of a finite system and an infinite system. In the former case, statement (2) is wrong because the assumption in statement (2b) is wrong. In the latter case, statement (2) is wrong because the conclusion in statement (2a) is wrong (as it has been established by Coombes and Laue).
Now, their position could in a sense be considered perfectly logical; we can adopt whatever definition we want, and here they accepted that by definition the temperatures of bodies in equilibrium with one another are equal—even if their mean-molecular-kinetic-energy values are not. But by presenting that paragraph as a “conclusion” they gave the impression their letter’s preceding math somehow compelled that choice.
The Non Sequitur
If this is what Velasco et al. intended then their rejection of the Ideal-Gas Law definition is a non sequitur. All ten of their letter’s equations are perfectly consistent with the Ideal-Gas Law. The first four deal with the infinite-limit case to which Coombes & Laue’s paper was directed, the next four deal with the finite case but don’t include temperature T, the ninth just defines the limit the Coombes & Laue paper tacitly assumed, and we will now see that the T value given by the tenth equation for the finite case is merely an overall average entirely consistent with the Ideal-Gas Law; it tells us nothing about the gradient in local temperature.
All Velasco et al.’s letter says about Equation 10 is, “Indeed, we have shown (see (41) in Román et al (1995)) that, in the microcanonical ensemble, the temperature of the system under consideration is given by
which remains finite in the thermodynamic limit.”
In that equation f = 3 for a monatomic gas, N is the total number of molecules, and E is the molecules’ total (potential + kinetic) energy. So the value implicitly given for T is obviously proportional to the average total energy E/N. And so long as total energy is partitioned 2:3 between potential and kinetic energy the resultant T value is the same as the one the Ideal-Gas Law gives for average temperature.
So Equation 10 is entirely consistent with the reason (2b) assertion that temperature is proportional to mean molecular kinetic energy. And, again, it tells us nothing about the lapse rate, which is the gradient in local temperature.
Derivation of Equation 10
Nor is anything in the Román et al. paper’s derivation of Velasco et al.’s Equation 10 inconsistent with statement (2b). Velasco et al.’s Equation 10 is Román et al.’s Equation 41, which was derived in an impressive feat of calculus from the following rather forbidding Equations 4 and 40:
The reader is invited to skip those equations initially but refer back to them as we explain that they implement a previously-mentioned statistical-mechanics definition of temperature as a quantity proportional to the reciprocal of the rate dS/dE at which system entropy S increases with system energy E.
As we observed above, entropy is simply proportional to the total number of possible states if all states are equally probable. In the case of our ideal-gas column each state is completely specified by a point in a 6N-dimensional “phase space” in which each of the 6N dimensions is the x, y, or z component of a corresponding molecule’s position q or momentum p. Also, the states are not discrete if we stick as Román et al. did to classical mechanics, and according to Liouville’s Theorem equal “volumes” in this phase space are equally likely.
So to arrive at a quantity from whose logarithm the entropy could be inferred Román et al. performed the continuous equivalent of counting states: they integrated through the phase space as Equation 4 above indicates to obtain the volume Φ of the portion of phase space where the molecules’ potential and kinetic energies sum to no more than some total value E. Entropy S is therefore proportional to the logarithm of Φ. So, as we said above, Equation 40 (in which Ω ≡ ∂Φ/∂E) implements the statistical-mechanics definition of temperature as proportional to the reciprocal of dS/dE.
Fortunately, the non-trivial calculus by which Román et al. obtained their Equation 41 and thus Velasco et al.’s Equation 10 from Román et al.’s Equation 40 need not detain us, because Equation 4 already reveals that Φ results from integrating altitude (which is every third q component) out of the expression. As we set out to show, therefore, the temperature T Román et al. obtained by differentiating the logarithm of Φ is an average over all altitudes, not a local quantity from which lapse rate can be inferred. And nothing in their math implies that local temperatures uniformly equal that average temperature. So it was misleading for Velasco et al. to present as a “conclusion” their assertion that “the assumption in statement (2b) is wrong.”
Their actual reasoning was probably as follows. Statement (1) is in essence the Zeroth Law, so they felt they had to insist on it and therefore reject statement (2). Having found a non-zero kinetic-energy gradient, however, they were forced to accept statement (2a), so contending that statement (2b) is wrong may have seemed to be their only option. To declare their loyalty to the Zeroth Law, in other words, they thought they needed to reject the Ideal-Gas Law.
But they could instead have viewed the Zeroth Law as an approximation that doesn’t rule out so small a lapse rate. According to that view rejecting the Ideal-Gas Law would amount to enforcing the Zeroth Law in a domain where its writ doesn’t run.
Science Is Hard
Again, there is no practical significance to whether that lapse rate would be exactly zero or only so small that it may as well be. But equilibrium-lapse-rate discussions that occurred around a decade ago at sites like Science of Doom, Tallbloke’s Talkshop, Climate Etc., and Watts Up with That reveal something that we laymen may profit from keeping in mind when we’re told what “scientists say” or that a paper has been peer-reviewed by scientists: science is hard, even for scientists.
In those discussions a number of PhD scientists (as well as laymen such as yours truly) struggled with a question to which Velasco et al.’s Equation 8 seems to provide a clear answer. That equation clearly says that at equilibrium the kinetic-energy gradient imposed by a gravitational field on a finite ideal gas is non-zero (albeit, for any significant quantity of gas, incomprehensibly small). And its derivation appears to be based on no assumptions other than the basic axioms of statistical mechanics. No critic we’re aware of has identified any errors in that derivation, we have found no math errors of any consequence, and Equation 8 checks out on the simple numerical examples we’ve been able to calculate.
Yet despite that unambiguous math and many disputants’ credentials the discussion was replete with ambiguities and confusion. To illustrate this we’ll return to Robert G. Brown, the Duke University physicist whose “Refutation of Stable Thermal Equilibrium Lapse Rates” we described in the previous post. Without identifying any purported error in Velasco et al.’s or Román et al.’s math Dr. Brown responded to an observation about Velasco et al.’s derived kinetic-energy gradient by stating that “Velasco et al is almost certainly wrong in their conclusion.” In that response he seemed implicitly to treat the thermodynamics and Ideal-Gas Law definitions of temperature as equivalent.
Dr. Brown’s reasoning was based on the thought experiment set forth in his “Refutation”: opposite ends of a vertically extending silver (i.e., high-thermal-conductivity) wire are thermally coupled at corresponding altitudes to the erstwhile-isolated ideal-gas column. If Velasco et al. were right that before coupling the gas column’s equilibrium molecular-kinetic-energy gradient would not be exactly zero, Dr. Brown reasoned, then the similarly isolated wire’s could be non-zero, too. And in any event the rates in the column and the wire before coupling wouldn’t likely be equal. But “if a gas has a lapse rate and metal has a different one then you have perpetual motion.” (Note that in calling Velasco et al.’s kinetic-energy gradient a lapse rate—i.e., a temperature gradient—Dr. Brown tacitly adopted temperature’s Ideal-Gas Law definition.)
In reaching his conclusion Dr. Brown apparently assumed that the equilibrium gradients would be the same after coupling as they had been before, so in the post-coupling equilibrium state a temperature difference at the wire-to-gas junctions would—perpetually—drive heat into the wire at one end and out of it at the other. Since this would be an “implicit violation of the second law,” he contended, Velasco et al. must be “almost certainly wrong.”
But the assumption that coupling wouldn’t change the equilibrium gradients ignores how Velasco et al. arrived at the gradient that Equation 8 implies. In essence the way in which they arrived at that equation involved “counting” only the gas-column microstates that have some given total-energy value. However, that same-total-energy constraint gets relaxed by coupling, which allows energy to flow back and forth between the gas and the wire. So the fact that the gradients differ in isolation doesn’t necessarily mean they’d differ after coupling.
Even if they did, moreover, how could Dr. Brown be sure that Fourier’s Law is so exact as to require heat flow no matter how small the non-zero temperature difference might be at the gas-wire interface? “Note,” he wrote, “that the second law doesn’t care about how large the violation is — if it is macroscopically violated you can, in principle, work free-lunch magic.” But is that really true if we’re using Ideal-Gas Law definition of temperature? What if under that definition Fourier’s Law is exact only in the infinite-number-of-molecules limit?
Consider, for example, the opposite extreme: only two molecules. Let’s say they’re traveling in opposite directions along the same line toward a head-on collision. If one molecule’s mass is twice the other’s, its speed with respect to their center of mass is half the other’s, so before collision the less-massive molecule has twice the more-massive one’s kinetic energy. Under the Ideal-Gas Law definition of temperature Fourier’s law would say that the body with more molecular kinetic energy per molecule tends to transfer energy to the body with less. Will the collision therefore transfer some of the less-massive molecule’s kinetic energy to the more-massive one?
Conservation of momentum tells us no; the two molecules’ velocities will simply be reversed, so their kinetic energies will remain unchanged. This suggests that Fourier’s Law is merely approached as the number of molecules gets large and may therefore be exact only in the infinite limit. So in rejecting Velasco et al.’s result Dr. Brown may have conflated different temperature definitions.
Now, we hasten to add that Dr. Brown wrote thousands of words in response to scores of questions about the lapse-rate topic, and what we quoted were just blog comments, so it’s entirely possible that he didn’t intend the meanings we attributed to him. But we think those passages are fairly representative of the type of confusion such topics can cause even among scientists.
Conclusion
Whether the equilibrium lapse rate would be exactly zero or just very close depends of one’s definition of temperature. The different definitions’ results don’t differ as a practical matter, but the resultant confusion among credentialed disputants should serve as a reminder to us laymen that science is hard even for scientists.