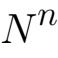Introduction
This post and the next will be directed to a highly academic question. The question concerns a thermally isolated ideal gas subjected to a uniform gravitational field. More specifically, it deals with the gas’s equilibrium lapse rate—i.e., the rate at which temperature decreases with altitude after the gas has so settled that there’s no macroscopic flow of air or heat. The question is whether that rate would equal zero exactly or just be so small that it might as well equal zero.
Real-world lapse rates play a large role in what’s often called the “greenhouse effect” and thus in global-warming discussions. But the reader is forewarned that our question about the equilibrium lapse rate has no actual climate implications. Indeed, the difference we’ll discuss between exactly and almost zero would be too small to measure. So the reader might be justified in likening the question to how many angels can dance on the head of a pin.
The equilibrium lapse rate has nonetheless been the subject of thousands of words of debate on many climate blogs. And those debates have sometimes been complicated by a failure to keep in mind what these posts’ discussion will emphasize: that the term temperature has more than one definition and that there’s a sense in which thermodynamics is just an approximation—albeit an exceedingly good one.
This first post will therefore explain why according to the Ideal-Gas Law an equilibrium lapse rate that’s exactly zero in a gravitational field would be inconsistent with energy conservation. The second post will describe the confusion that can arise because thermodynamics nonetheless defines it to be zero.
A Plausible Theory
In most locations the lapse rate we encounter when we, say, ascend a mountain is on the order of 6.5°C/km. The lapse rate’s major cause is that uneven heating makes surface air warmer and less dense at some locations than at others, so nearby cooler, denser air displaces warmer air parcels upward. As a result a type of cooling occurs that in a sense is complementary to the type of heating we may notice in a bicycle pump.
In the case of a bicycle pump the pumped air’s temperature increases because the pump converts the user’s air-compression work to heat. In the case of the atmosphere a rising air parcel’s temperature drops because lower pressure aloft allows it to expand, and the parcel loses heat energy to the work its expansion performs on the surrounding air. That expansion is often called adiabatic expansion because air doesn’t conduct heat well enough for conduction from the surrounding air to have much effect on the rising air’s cooling.
Atmospheric physicists widely accept that such adiabatic expansion is largely responsible for the lapse rate. But some scientists and others believe that a substantial lapse rate would result even if uneven heating and the resultant adiabatic expansion did not occur.
Imagine an ideal (non-radiative) gas, they say, that a non-heat-conducting tube extending vertically in a uniform gravitational field so isolates from the sun and other heat sources and sinks as to prevent any heating or cooling, uneven or not. They contend that even at equilibrium the conversion that occurs between individual molecules’ potential and kinetic energies would cause a lapse rate comparable in magnitude to those we observe in the actual, turbulent atmosphere.
Specifically, a gas molecule falling in a gravitational field gains kinetic energy, whereas a rising molecule loses it. Temperature according to the Ideal-Gas Law we learned in high-school physics is a measure of those molecules’ average translational kinetic energies, so proponents of this theory conclude that even without adiabatic expansion the gas molecules’ loss of kinetic energy as they rise would result in lower temperatures at higher altitudes.
A Purported Refutation
Although that theory sounds plausible, statistical mechanics proves that as a practical matter any resultant lapse rate would be too small to measure. But statistical mechanics isn’t everyone’s cup of tea. So Duke University physicist Roger G. Brown’s 2012 “Refutation of Stable Thermal Equilibrium Lapse Rates” provided a more-accessible explanation in the form of a proof by contradiction. As we will see, however, his conclusion was not quite accurate.
Dr. Brown assumed for the sake of argument that the lapse rate imposed by a uniform gravitational field would indeed be non-zero: there would be a temperature difference between the gas column’s top and bottom. As far as the proof goes, he wrote, “[t]he magnitude of the difference, and the mechanism proposed for this separation are irrelevant. . . .”
Whatever its magnitude a non-zero value would according to Dr. Brown present us only two choices. One is that the gravitational field would somehow keep the lapse rate constant and thereby cause heat to flow at a constant rate from the column’s bottom to its top. But he argued that this would imply an impossibility: one could achieve perpetual motion by connecting a heat engine between the gas column’s top and bottom.
Since perpetual motion is impossible, he said, we’re left with the other choice: that the heat flow would eventually eliminate the temperature difference and thus the heat flow it caused. In other words, the non-zero lapse rate assumed for the sake of argument is not an equilibrium condition. Q.E.D.
Thermodynamically, that refutation seems unassailable; Fourier’s Law seems to impose no limit on its rule that heat flow is proportional to the temperature gradient. And for all practical purposes Dr. Brown’s conclusion is indeed correct. But as a theoretical matter his insistence that the temperature difference’s magnitude is irrelevant makes his conclusion inconsistent with what we know about ballistic motion.
Consider the degenerate case in which our “gas” column consists of only a single, monatomic molecule. Let’s say that when the molecule is located at the bottom of the column its kinetic energy has the value E = 3kT/2 associated with some temperature T, where k is Boltzmann’s constant. Conversion from kinetic energy to potential energy as the molecule rises causes the former to decrease with altitude at a rate mg, where m is the molecule’s mass and g is the acceleration of gravity. Consequently, the molecule’s kinetic energy will be zero—i.e., the “temperature” of the “gas” will be absolute zero—at the altitude z = E/m/g at which the molecule’s potential-energy increase mgz equals its altitude-zero kinetic energy E.
The lapse rate is therefore the ratio T/z that the zero-altitude temperature bears to the zero-temperature altitude. For a monatomic molecule whose molecular weight is air’s average, that works out to about 23°C/km. This is a substantial lapse rate.
Now, faced as he was with that objection, Dr. Brown might perhaps have been justified in dismissing it as a quibble. Note, he might have argued, that the absolute-zero-temperature altitude z in our lapse-rate formula T/z is proportional to the total kinetic energy over all the molecules. At standard temperature and pressure even just a milliliter of gas contains billions of billions of molecules, so z would be astronomical; in a macroscopic gas the equilibrium lapse rate would as a practical matter be too small to measure. And even in principle the random variations caused by molecular motion could make the required measurement time prohibitively long.
But that wasn’t his response; he didn’t concede that the equilibrium lapse rate doesn’t have to be exactly zero. Instead he pointed out that a single molecule or a small number of them can’t for most purposes be considered a gas. Although that’s a reasonable enough observation it doesn’t support his position unless when there are enough molecules to be called a gas the lapse rate somehow suddenly stops being merely small and starts being exactly zero.
Coombes and Laue
Anyway, we will now show that in a finite gas an equilibrium lapse rate that’s exactly zero would violate conservation of energy. To that end we will paraphrase a plausibility argument made by Coombes & Laue for the opposite proposition: that kinetic-energy loss by rising molecules is not inconsistent with a uniform temperature.
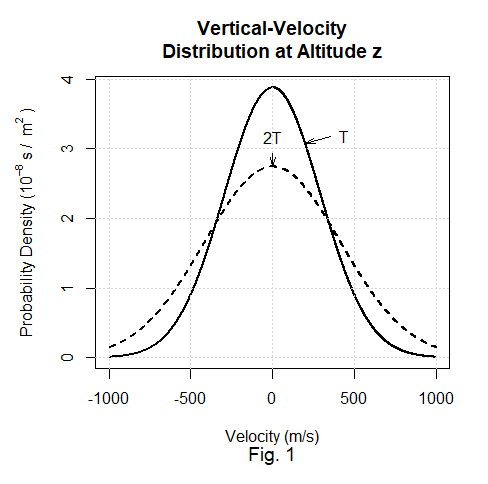
In their argument Coombes & Laue employ the density f(v, z) of the probability that in a gravitational field a randomly chosen ideal-gas molecule will have a vertical-velocity component v and an altitude z. They start with the assumption that there’s at least one altitude z at which the molecular speed in three dimensions has the Maxwell-Boltzmann distribution associated with some temperature T. Being a chi distribution for three degrees of freedom, the Maxwell-Boltzmann speed distribution implies a Gaussian distribution of the vertical-velocity component. At some particular altitude z, that is, Coombes & Laue assume that
where C is a coefficient proportional to the molecular density at that altitude z.
Fig. 1 illustrates the distribution’s temperature dependence: a higher temperature means a greater velocity variance.
Making the simplifying assumption that molecules don’t collide, Coombes & Laue followed the consequently uninterrupted rising and falling molecules to demonstrate that the assumption of a given-temperature Maxwell-Boltzmann distribution at one altitude implies the same-temperature Maxwell-Boltzmann distribution at every other altitude.
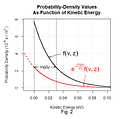
To visualize their approach we plot f for a selected altitude not against those rising molecules’ vertical velocities v but rather against the kinetic energies those velocities imply:
Making that change of variable enables us to impose a common kinetic-energy loss on all the molecules by merely shifting the curve leftward. So a leftward shift by the mgΔz kinetic-energy loss the molecules suffer when they rise by Δz gives us the distribution at z + Δz.
Since less average molecular kinetic energy means a lower temperature, one might be tempted to conclude that such an energy loss would mean a lower temperature at higher altitudes. As the dashed portion of Fig. 2’s red curve indicates, however, the altitude change culls the molecules whose kinetic energies at z weren’t great enough for them to reach z + Δz. The remaining kinetic energy is therefore shared among fewer molecules.
Moreover, that change of variable converted the curve from Gaussian to exponential, and it is a property of exponential functions that the ratio of the shifted curve to the original is identical at all argument values. In other words, normalizing the two curves would yield identical distributions: the temperatures are the same at both levels and by extension at all levels.
The Exponential Assumption
In short, Coombes & Laue showed that ballistic molecular motion is not inconsistent with a zero lapse rate. But the thing that makes gravitational culling keep the temperature uniform in their argument is the assumption that the vertical-velocity distribution is exponential in the associated kinetic energy. If that function is not exactly exponential, then the lapse rate is not exactly zero.
And in a finite gas that function can’t be exactly exponential. Although an exponential function can decay as its argument increases, it never quite reaches zero. So if the vertical-velocity distribution were exactly exponential there would be a non-zero probability of molecular kinetic energies that exceed the gas’s total energy.
That’s obviously impossible. So Coombes & Laue’s result is exact only in the limit of an infinite number of molecules. And Dr. Brown’s insistence that the temperature difference’s magnitude is irrelevant therefore undermines his proof.
Conclusion
Yet Fourier’s Law seems to say the equilibrium temperature difference has to be zero, and many if not most physicists would agree. Our next post will therefore explore some of the reasons why they would. We will see that those reasons involve the nature of thermodynamics as well as what we mean by temperature.